Proof of illegal kickbacks: Doctor admits he was compensated by opioid manufacturer for issuing unnecessary prescriptions
10/29/2017 / By Russel Davis

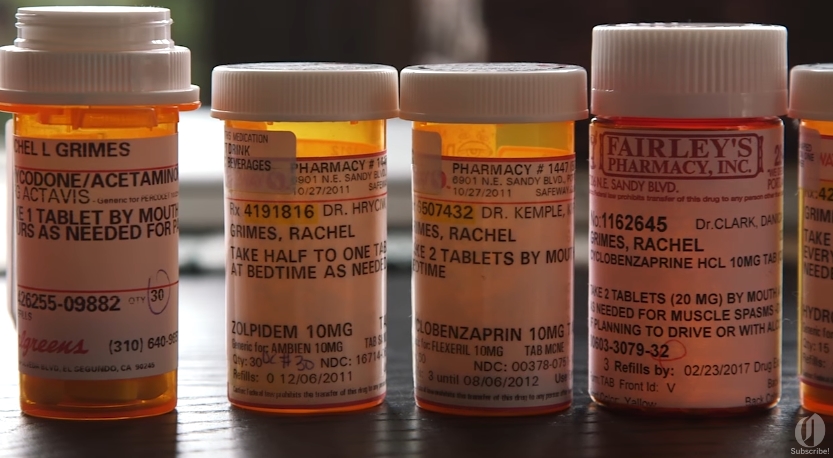
A Rhode Island-based physician has recently admitted to participating in a fraudulent scheme and accepting hundreds of thousands of dollars in kickbacks to illegally prescribe fentanyl, a highly-addictive opioid. A Reuters report has revealed that Dr. Jerrold Rosenberg, 36, has pleaded guilty before the U.S. District Court in Providence on grounds of health care fraud and illegally prescribing the addictive fentanyl-based cancer pain drug Subsys. The drug, an under-the-tongue spray, is manufactured by Chandler, Arizona-based Insys Therapeutics Inc.
According to the prosecutors, Rosenberg participated in the fraudulent scheme from 2012 to 2015 and had received a whopping $188,000 in kickbacks. The prosecutors have reported that the kickbacks have been in the form of speaker fees from Insys. The prosecutors also noted that the speaker fees became the doctor’s basis in prescribing the opioid spray. Likewise, the reports have indicated that the physician fraudulently claimed that the patients suffered from cancer pain when the prescriptions are not given, thus securing insurance approvals for Subsys. (Related: Doctors are prescribing DEADLY fentanyl for cash and lavish dinners, reveals report.)
The doctor has agreed to settle $754,736 in restitution to healthcare benefit programs as part of the plea agreement. Aside from this, the physician is facing a maximum prison sentence of 15 years and is slated to be sentenced on Jan. 16. The doctor’s lawyer has declined a request for comment.
The probe against the drug maker comes during a national opioid abuse epidemic. Federal prosecutors in Boston have charged six former Insys executives and managers — which included ex-Chief Executive Michael Babich — in December on grounds of performing a scheme that bribed physicians into prescribing Subsys and defrauding insurers. All the six former officials have pleaded not guilty before the court. Likewise, several other states have filed lawsuits against other former company officials and medical practitioners that have prescribed the fentanyl-based spray.
In addition, the pharmaceutical company faces charges filed by attorneys general in Arizona and New Jersey. The company has been reported to have settled up to $9.45 million to resolve probes by attorneys general in certain states including Oregon, New Hampshire, Illinois, and Massachusetts. The drug firm has been allegedly coordinating with the U.S. Justice Department in order to resolve issues associated with the misdeeds of former employees.
“We have taken necessary and appropriate steps to prevent past mistakes from happening in the future and are committed to conducting business according to high ethical standards and the interests of patients. We also continue to work with relevant authorities to resolve issues related to the misdeeds of former employees,” an Insys spokesperson has told Daily Mail online.
Opioid abuse becoming more prevalent across the U.S.
The Centers for Disease Control and Prevention has noted that opioid abuse has resulted in more than 33,000 deaths in 2015 alone. The federal agency has also cautioned that the rates have been continuously increasing. According to the American Society for Addiction Medicine (ASAM), the drugs are chemically related and work with the opioid receptors on nerve cells in the brain and nervous system in order to alleviate pain and generate pleasurable effects.
Data published by the ASAM have also shown that opioid abuse has already reached the youth. The data revealed that as many as 276,000 adolescents aged 12 to 17 years old are current non-medical users of pain relief drugs in 2015. Nearly half of these teens have reported being addicted to painkillers, the ASAM data has shown. The report has also indicated that the number of opioid prescriptions among teens have nearly doubled between 1994 to 2007.
Sources include:
Tagged Under: addiction, Big Pharma, bribery, corruption, doctors, drug addiction, drug cartels, drug dependence, kickbacks, Opioid, opioid addiction, painkillers